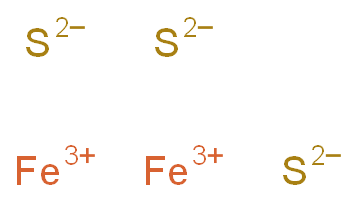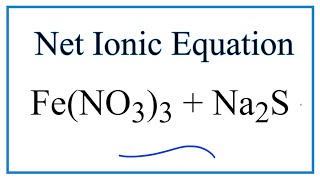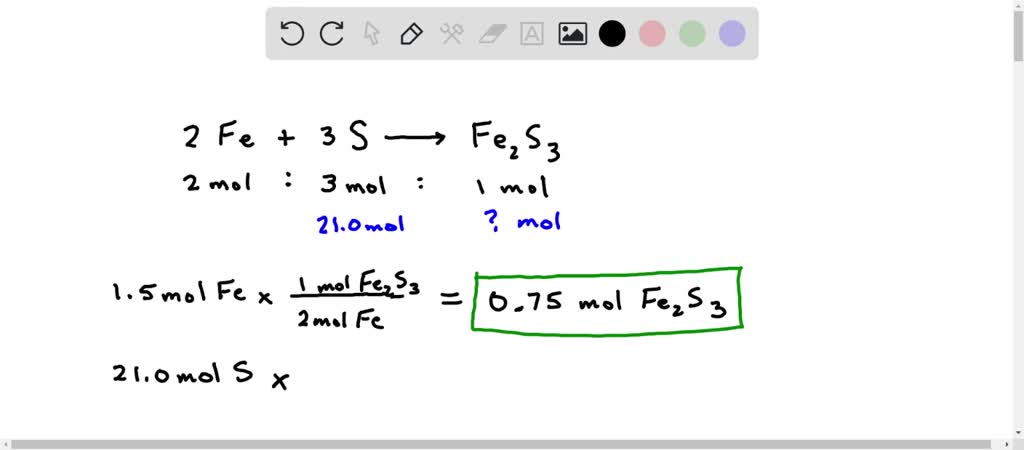
SOLVED: 1A. Consider the balanced chemical equation that follows: 2Fe(s) + 3S(s) → Fe2S3(s) This equation can be read as 2 moles of Fe + 3 moles of S → 1 mole

If the reaction occurs between Fe2S3(s) and H+ (aq) and the equation is properly balanced with the - Brainly.com

for the reaction fe2s3+5o2gives raise to 2feso4+so2 the equivalent weight of fe2s3 is - Chemistry - Solutions - 13840395 | Meritnation.com

How to balance Fe+S8=Fe2S3|Chemical equation Fe+S8=Fe2S3|Reaction balance Fe+S8=Fe2S3| Fe+S8=Fe2S3 - YouTube

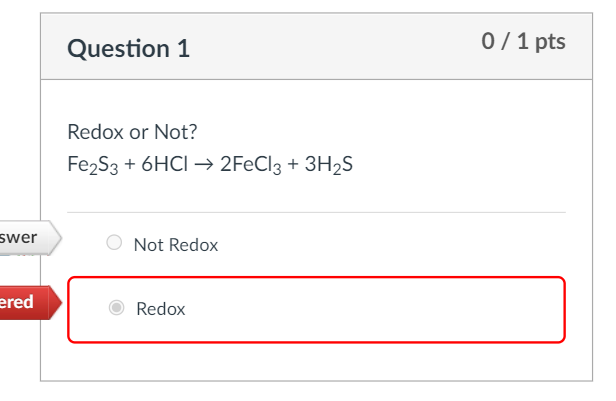
SOLVED: Balance the equation Fe2S3(s) → Fe(s) + S(s) + 69kJ 1b) 2A. How many moles of sulfur are in one mole of Fe2S3(s)? 2b. How many moles of Sulfur is in